Efficacy and Safety of Denosumab for the Treatment of Osteoporosis a Systematic Review
Introduction
Osteoporosis is defined as a bone illness that is characterized by decreased bone mineral density (BMD) and deteriorated micro-compages of the skeleton, causing fragile bones and fracture risk(Brown et al., 2006). Osteoporosis is mutual, with a prevalence of 20–40% in postmenopausal women and vi–8% in men ≥50ºyears of age (Dubois et al., 2002; Sharma et al., 2008; Hernlund et al., 2013; Lee et al., 2013; Cosman et al., 2014; Institute for Clinical Systems Improvement (ICSI), 2017). It is more than common in postmenopausal women, persons ≥65ºyears of age, Caucasians, Asians, and persons with a small body frame (Cosman et al., 2014; Institute for Clinical Systems Improvement (ICSI), 2017). Osteoporosis can occur as part of the aging process or secondarily due to nutritional deficiency, metabolic disorders, or medication side effects (National Library of Medicine, 2010; Cosman et al., 2014). Certain endocrine, gastrointestinal, hematologic, autoimmune, and central nervous organisation (CNS) disorders increase the risk of osteoporosis. Medications such as long-term anticoagulation (Caraballo et al., 1999; Gage et al., 2006), hormonal therapies (Tit et al., 2017; Tit et al., 2018), glucocorticosteroids (Etminan et al., 2008; Loke et al., 2011), some immunosuppressants (Anastasilakis et al., 2019), lithium (Zamani et al., 2009), thiazolidinediones (glitazones) (Majumdar et al., 2016), selective serotonin reuptake inhibitors (Diem et al., 2007; Richards et al., 2007), and long-term proton pump inhibitor employ (Ngamruengphong et al., 2011; Khalili et al., 2012) may besides cause osteoporosis(Cosman et al., 2014; Institute for Clinical Systems Improvement (ICSI), 2017). Osteoporosis accounts for greater than 90% of hip and vertebral fractures in women aged 65–84ºyears (NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy, 2001), utilizing major health services worldwide (Prevention and management of osteoporosis, 2003; Burge et al., 2007). With increment in the average life span, postmenopausal osteoporosis is condign a serious public health result in China and in many other countries. Moreover, osteoporotic fracture causes permanent disability, admission to institutional care, and even decease (Sambrook and Cooper, 2006).
Currently, lifestyle recommendations (vitamin D and calcium supplementation, practise, and smoking alcohol abeyance) and antiresorptive agents equally standard therapies for osteoporosis, with bisphosphonates as get-go-line treatment, were proved to have beneficial effects on BMD and risk of frail fractures in postmenopausal women (Adachi et al., 2009; Clarke, 2009; Holick et al., 2011; Bauer, 2013; Cosman et al., 2014; Plant for Clinical Systems Improvement (ICSI), 2017; Qaseem et al., 2017). However, fracture occurs if the treatment strategies did not yield acceptable response. Therefore, additional effective treatment agents should be identified to improve the prognosis of osteoporosis.
With advances in os physiology, RANKL has already been identified as an important os remodeling mediator. Moreover, RANK, every bit a receptor of RANKL, is observed during several stages of differentiation on osteoclast surface (Boyle et al., 2003; Hofbauer and Schoppet, 2004). The RANKL–RANK interaction is controlled by the soluble cytokine receptor, osteoprotegerin, which sequesters RANKL and neutralizes its furnishings (Simonet et al., 1997; Lacey et al., 1998).
Denosumab, a fully human monoclonal IgG2 antibiotic, binds to RANK ligand and affects the formation, function, and survival of osteoclasts (Fuller et al., 1998; Yasuda et al., 1998; Lacey et al., 2000). Several randomized controlled trials (RCTs) have been conducted for evaluating the handling efficacy and safety of denosumab in osteoporosis or low-BMD postmenopausal women. Just the treatment effectiveness of denosumab should be summarized to report the magnitude of effect estimates.
Therefore, this report aimed to investigate the treatment efficacy and safety of denosumab vs. placebo in osteoporotic or low-BMD postmenopausal women based on RCTs.
Methods
Information Sources, Search Strategy, and Pick Criteria
The Preferred Reporting Items for Systematic Reviews and Meta-Analysis Statement guidelines were used to conduct this report (Moher et al., 2009). PubMed, Embase, Cochrane library, and ClinicalTrials.gov were explored for published articles from inception till May 2019. The following search terms were used to recollect the articles: osteoporosis, postmenopause, postmenopausal, women, denosumab, and randomized controlled trials. To obtain more than appropriate and highly accurate studies, the reference lists of the obtained articles were also reviewed.
Two researchers selected the manufactures later initial screening. After that, careful screening of titles and abstracts of these articles was done. If the study was considered relevant, then total text of the study was obtained. Eligible studies should meet the following inclusion criteria (Chocolate-brown et al., 2006): written report blueprint: studies designed as RCTs (Hernlund et al., 2013); patients: osteoporotic or low-BMD postmenopausal women (Lee et al., 2013); intervention: denosumab (Dubois et al., 2002); control: placebo (Sharma et al., 2008); outcomes: BMD or fracture at various sites, and any other potential adverse events. If several publications were available with increasing number of patients or longer follow-up for the same grouping, simply data of 1- to iii-year follow-upward duration were used for statistical analysis.
Data Collection and Quality Assessment
The full-text and relevant data extraction from each written report into the coding table in Microsoft Excel software was conducted by ii reviewers. The following information including the first authors' surname, publication yr, country, sample size, mean age, body mass alphabetize (BMI), disease status, intervention and control, follow-up elapsing, and reported outcomes was extracted from each study. The JADAD scale was used to assess the quality of enrolled studies, which is based on randomization (i or 0), concealment of the treatment allotment (1 or 0), blinding (1 or 0), completeness of follow-up (1 or 0), and the apply of intention-to-treat assay (ane or 0), and the scale system ranged from 0 to 5 (Jadad et al., 1996). The data drove and quality cess were performed by two independent authors, and any conflicts betwixt them were settled by an additional author past reviewing the original article.
Statistical Analysis
Effect estimates for continuous information were presented as weighted mean differences with its 95% conviction intervals (CIs), while those of chiselled data were expressed equally relative risks (RRs) with corresponding 95% CIs. If the data in individual studies were expressed equally median and range, and so the data were converted to estimated means ± standard deviation before assay. The summary event estimates for efficacy and safe profiles were evaluated using the random-furnishings model (DerSimonian and Laird, 1986; Ades et al., 2005). Heterogeneity was evaluated across the included studies using I-square statistics and p value for Q statistics, and I-foursquare greater than 50% or p < 0.10 was considered as significant heterogeneity (Higgins et al., 2003; Deeks et al., 2008). Sensitivity analysis was calculated to assess the impact of unmarried individual trial from the overall analyses for clinical fractures (Pedroza-Tobías, 1999). Subgroup analyses for BMD and fractures were conducted based on the sites, and the treatment furnishings of denosumab among various sites were calculated using an interaction test (Altman and Bland, 2003). Publication bias for clinical fractures was evaluated using funnel plot (a blueprint distribution roughly with the shape of a funnel indicates no publication bias), Egger (Egger et al., 1997), and Begg tests (Begg and Mazumdar, 1994). The inspective level for pooled results was two-sided, and p < 0.05 was regarded equally statistically significant. The analyses in this study were carried out through STATA software (version 12.0; Stata Corporation, College Station, TX, Us).
Results
Literature Search
A preliminary initial search yielded 971 related records. After the titles and abstracts were reviewed, 918 studies were excluded due to duplications or irrelevant topics. For the remaining 53 articles, full texts were obtained and and so reviewed. Of these, 42 were excluded for the following reasons: they used other command agents (n = 23), studies reported the same population (n = sixteen), and they were with no desirable outcomes (n = 3). Manual searching of the reference lists of the remaining studies yielded no boosted report. Finally, 11 RCTs were selected for conducting this meta-analysis (Table i; McClung et al., 2006; Os et al., 2008; Ellis et al., 2008; Cummings et al., 2009; Seeman et al., 2010; Bone et al., 2011; Kumagai et al., 2011; Nakamura et al., 2012; Nakamura et al., 2014; Gnant et al., 2015; Koh et al., 2016). Figure i represents a flowchart of the choice process, and inclusion and exclusion criteria.
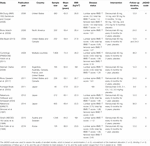
TABLE 1. Baseline characteristics of studies included in the meta-assay.
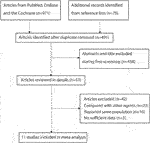
Effigy 1. Flow diagram of report identification, and inclusion and exclusion criteria.
Study Characteristics
11 RCTs recruited a total of 12,013 osteoporotic or low-BMD postmenopausal women. The follow-up elapsing for participants ranged from 9 to 36ºmonths, and 40–7,808 patients were included in each trial. The mean historic period of enrolled patients ranged from 57.6 to 72.3ºyears, and BMI ranged from 22.three to 27.8ºkg/mtwo. Six studies were conducted in Western countries, 4 studies in Eastern countries, and the remaining ane study in multiple countries. Ii trials had a score of 5, 8 trials had a score of 4, and the remaining one trial had a score of 3.
Bone Mineral Density
The summary results regarding the effectiveness of denosumab vs. placebo on BMD at various sites are presented in Figure 2. Overall, the results showed that the percentage of modify in BMD was significantly increased with denosumab when compared with placebo at 1/3 radius (WMD: 3.43; 95%CI: 3.24 to 3.62; p < 0.001), femoral cervix (WMD: three.05; 95%CI: i.78 to 4.33; p < 0.001), lumbar spine (WMD: 6.25; 95%CI: 4.59 to 7.92; p < 0.001), total hip (WMD: 4.36; 95%CI: 4.07 to 4.66; p < 0.001), trochanter (WMD: six.00; 95%CI: v.95 to six.05; p < 0.001), and total torso (WMD: 3.20; 95%CI: two.03 to 4.38; p < 0.001). The included studies showed a pregnant heterogeneity for BMD at 1/3 radius, femoral neck, lumbar spine, full hip, and total torso.

Effigy two. Summary results of BMD at diverse sites.
Fracture
The breakdown of the number of trials available for clinical fractures, nonvertebral fractures, vertebral fractures, and hip fractures was vi trials, 3 trials, three trials, and ane trial, respectively. The summary RRs indicated that the run a risk of clinical fractures (RR: 0.57; 95%CI: 0.51 to 0.63; p < 0.001), nonvertebral fractures (RR: 0.83; 95%CI: 0.70 to 0.97; p = 0.018), vertebral fractures (RR: 0.32; 95%CI: 0.25 to 0.forty; p < 0.001), and hip fractures (RR: 0.61; 95%CI: 0.37 to 0.98; p = 0.042) was significantly reduced in patients who received denosumab (Figure three). The included trials showed no heterogeneity for clinical fractures, nonvertebral fractures, and vertebral fractures. The results of sensitivity analysis indicated that the pooled conclusion for clinical fracture was stable and was unaltered by excluding whatever particular trial (Figure four). Finally, no pregnant publication bias was detected through clinical fracture information (p value for Egger: 0.742; p value for Begg: 0.707; Figure v).

Figure iii. Summary results of fracture risk at diverse sites.

Effigy 4. Sensitivity analysis of clinical fractures.
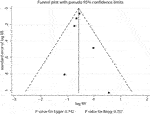
FIGURE 5. Funnel plot of clinical fractures. A pattern distribution roughly with the shape of a funnel indicates no publication bias.
Safety Profiles
The summary results regarding the risk of agin events are summarized in Supplementary Tabular array S1. First, no significant differences were observed between denosumab and placebo for the risk of whatever adverse events (RR: 1.00; 95%CI: 0.99–1.01; p = 0.995; without show of heterogeneity), treatment-related adverse events (RR: 0.96; 95%CI: 0.75–1.23; p = 0.740; without bear witness of heterogeneity), withdrawal due to adverse events (RR: one.12; 95%CI: 0.84–ane.48; p = 0.435; without evidence of heterogeneity), and expiry (RR: 0.80; 95%CI: 0.59–1.07; p = 0.137; without prove of heterogeneity). Second, for adverse events that occur in at least 10% of subjects, complications such as constipation (RR: 1.53; 95%CI: one.01–2.32; p = 0.043), flatulence (RR: i.58; 95%CI: one.12–2.22; p = 0.008), pharyngolaryngeal hurting (RR: 3.02; 95%CI: 1.12–8.11; p = 0.029), and rash (RR: 3.00; 95%CI: 1.17–7.68; p = 0.022) were significantly increased in patients who received denosumab, whereas denosumab therapy was associated with low risk of falling (RR: 0.80; 95%CI: 0.66–0.97; p = 0.022) and periarthritis (RR: 0.17; 95%CI: 0.04–0.66; p = 0.010). No other significant deviation was detected for any specific agin events. Finally, denosumab and placebo showed no significant differences regarding the occurrence of serious adverse events or specific serious adverse events.
Discussion
The prevalence of osteoporosis is on the rise and has get a serious public health result of global concern, especially in postmenopausal women with advanced age, inducing greater fracture risk at various sites. The current study was conducted based on eleven RCTs with 12,013 osteoporosis or low-BMD postmenopausal women to evaluate the efficacy and safety of denosumab vs. placebo across a wide range of characteristics. The results of this study suggested that denosumab showed association with high percentage change in BMD at one/3 radius, femoral neck, lumbar spine, total hip, trochanter, and total torso. Moreover, the gamble of diverse types of fractures such equally clinical fractures, nonvertebral fractures, vertebral fractures, and hip fractures was significantly reduced in patients who received denosumab. Furthermore, denosumab did not yield additional risks on any adverse events, handling-related adverse events, withdrawal due to agin events, and deaths. Although denosumab and placebo treatments showed meaning differences, these results might vary as fewer number of trials were included. As for homogeneity, the results from studies with greater weight were like. Hence, even though the results from studies with lower weight were more variable, the overall results were still robust.
In that location are a large number of systematic reviews and meta-analyses conducted focusing on denosumab for treating postmenopausal women with osteoporosis or low BMD. Anastasilakis et al. (2009) conducted a meta-assay of three RCTs and showed a significant decrease in the bone markers and increase of lumbar and hip BMD afterward handling with denosumab, whereas no significant benefits were observed on the gamble of fracture, and increased the infection run a risk. von Keyserlingk et al. (2011) conducted a meta-analysis based on four RCTs, and reported significant reduction in the take chances of fracture without increasing agin events in postmenopausal women who received denosumab. Zhou et al. (2014) carried out a meta-assay of 11 RCTs and demonstrated a meaning reduction in the take a chance of nonvertebral fractures and additionally yielded serious adverse events related to infection in osteoporotic or low-BMD postmenopausal women after treatment with denosumab. The meta-assay conducted by Gu et al. (2015) was based on four RCTs, and the results revealed that the BMD was increased and the os turnover markers were decreased in postmenopausal women after treatment with denosumab, whereas no significant adventure for adverse events was observed. However, several RCTs have already been conducted regarding the topic, merely should reevaluate the magnitude regarding the treatment effectiveness of denosumab vs. placebo in osteoporotic or low-BMD postmenopausal women. Therefore, the current quantitative meta-analysis was conducted to update the efficacy and safety of denosumab vs. placebo in postmenopausal women with osteoporosis or low BMD.
The summary results indicated that the percentage change of BMD in the denosumab group was greater than that of BMD in placebo at 1/3 radius, femoral neck, lumbar spine, total hip, trochanter, and total body, and these results were consistent with previous meta-analyses (Anastasilakis et al., 2009; Gu et al., 2015). Moreover, these results from private trials reported similar conclusions, and the results of this study provided the magnitude of pooled results. Moreover, the current study suggested that the risk of clinical fractures, nonvertebral fractures, vertebral fractures, and hip fractures was significantly reduced in patients who received denosumab. The potential reasons for these conclusions could be due to denosumab action in inhibiting RANKL, preventing os resorption, increasing BMD, and reducing fracture hazard in osteoporosis or low-BMD postmenopausal women (Bekker et al., 2004; Lewiecki et al., 2007).
The summary results showed that denosumab therapy caused greater take chances of constipation, flatulence, pharyngolaryngeal hurting, and rash, whereas the risk of falling and periarthritis were significantly reduced. The potential reasons for this could be that denosumab could affect the immune organization of the patients taking it and is associated with these adverse events (Canalis, 2010; Moen and Keam, 2011). Moreover, the hazard of falling and periarthritis were reduced due to increased levels of BMD in osteoporotic or low-BMD postmenopausal women (Hita-Contreras et al., 2014; Burnett et al., 2017).
Although the electric current study provides comprehensive effectiveness results regarding denosumab in osteoporotic or low-BMD postmenopausal women, several limitations still existed and should be mentioned. First, the summary results for BMD at various sites were available only in few trials, inducing instability in the magnitude of BMD in the denosumab group. Second, the risk of most of the agin events was bachelor in smaller trials, and the ability might not be enough to detect the divergence between denosumab and placebo groups. Finally, publication bias remained inevitable since the analysis was conducted on published RCTs and that the bias against the publication of negative results is well known (Murad et al., 2018).
In determination, the findings of this meta-analysis indicated that osteoporotic or low-BMD women who received denosumab had increased BMD and reduced fracture risk at various sites. Moreover, the frequency of adverse events between denosumab and placebo groups was similar. Future large-scale RCTs that compare the treatment effectiveness of denosumab with other traditional drugs in patients at various stages should be conducted.
Author Contributions
YC carried out the studies, JZ and YZ participated in collecting data, and YC drafted the manuscript. JP and BW performed the statistical analysis and participated in its design. JZ and YZ helped to draft the manuscript. All authors read and canonical the final manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Fabric
The Supplementary Cloth for this commodity can be found online at: https://world wide web.frontiersin.org/manufactures/10.3389/fphar.2021.588095/total#supplementary-material.
References
Adachi, J. D., Kennedy, C. C., Papaioannou, A., Ioannidis, G., Leslie, W. D., and Walker, Five. (2009). Treating osteoporosis in Canada: what clinical efficacy information should be considered by policy decision makers? Osteoporos. Int. 20 (10), 1785–1793. doi:x.1007/s00198-009-0870-9
PubMed Abstract | CrossRef Total Text | Google Scholar
Ades, A. E., Lu, Thou., and Higgins, J. P. (2005). The interpretation of random-effects meta-analysis in decision models. Med. Decis. Making 25 (half dozen), 646–654. doi:10.1177/0272989X05282643
PubMed Abstract | CrossRef Full Text | Google Scholar
Anastasilakis, A. D., Toulis, M. A., Goulis, D. G., Polyzos, S. A., Delaroudis, S., Giomisi, A., et al. (2009). Efficacy and prophylactic of denosumab in postmenopausal women with osteopenia or osteoporosis: a systematic review and a meta-analysis. Horm. Metab. Res. 41 (x), 721–729. doi:ten.1055/due south-0029-1224109
PubMed Abstruse | CrossRef Total Text | Google Scholar
Anastasilakis, A. D., Tsourdi, E., Makras, P., Polyzos, Due south. A., Meier, C., McCloskey, E. V., et al. (2019). Bone disease post-obit solid organ transplantation: a narrative review and recommendations for management from the European Calcified Tissue Society. Bone 127, 401–418. doi:10.1016/j.bone.2019.07.006
PubMed Abstruse | CrossRef Full Text | Google Scholar
Bekker, P. J., Holloway, D. Fifty., Rasmussen, A. Southward., Spud, R., Martin, S. Westward., Leese, P. T., et al. (2004). A single-dose placebo-controlled study of AMG 162, a fully man monoclonal antibody to RANKL, in postmenopausal women. J. Bone Miner Res. 19 (7), 1059–1066. doi:x.1359/JBMR.040305
PubMed Abstract | CrossRef Full Text | Google Scholar
Bone, H. G., Bolognese, 1000. A., Yuen, C. M., Kendler, D. L., Miller, P. D., Yang, Y. C., et al. (2011). Furnishings of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with depression bone mass. J. Clin. Endocrinol. Metab. 96 (4), 972–980. doi:10.1210/jc.2010-1502
PubMed Abstract | CrossRef Total Text | Google Scholar
Os, H. G., Bolognese, M. A., Yuen, C. K., Kendler, D. L., Wang, H., Liu, Y., et al. (2008). Effects of denosumab on os mineral density and bone turnover in postmenopausal women. J. Clin. Endocrinol. Metab. 93 (half dozen), 2149–2157. doi:10.1210/jc.2007-2814
PubMed Abstruse | CrossRef Total Text | Google Scholar
Brown, J. P., Fortier, M., Frame, H., Lalonde, A., Papaioannou, A., Senikas, V., et al. (2006). Canadian Consensus Conference on osteoporosis, 2006 update. J. Obstet. Gynaecol. Can. 28 (2 Suppl. 1), S95–S112. doi:10.1016/s1701-2163(16)32087-four
PubMed Abstract | CrossRef Full Text | Google Scholar
Burge, R., Dawson-Hughes, B., Solomon, D. H., Wong, J. B., King, A., and Tosteson, A. (2007). Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J. Os Miner Res. 22 (three), 465–475. doi:ten.1359/jbmr.061113
PubMed Abstruse | CrossRef Full Text | Google Scholar
Burnett, W. D., Kontulainen, S. A., McLennan, C. E., Hazel, D., Talmo, C., Wilson, D. R., et al. (2017). Proximal tibial trabecular bone mineral density is related to pain in patients with osteoarthritis. Arthritis Res. Ther. 19 (1), 200. doi:10.1186/s13075-017-1415-ix
PubMed Abstruse | CrossRef Full Text | Google Scholar
Caraballo, P. J., Heit, J. A., Atkinson, E. J., Silverstein, K. D., O'Fallon, W. G., Castro, 1000. R., et al. (1999). Long-term utilise of oral anticoagulants and the risk of fracture. Arch. Intern. Med. 159 (xv), 1750–1756. doi:10.1001/archinte.159.15.1750
PubMed Abstract | CrossRef Total Text | Google Scholar
Cosman, F., de Beur, S. J., LeBoff, Chiliad. S., Lewiecki, E. M., Tanner, B., Randall, S., et al. (2014). Clinician's guide to prevention and handling of osteoporosis. Osteoporos. Int. 25 (ten), 2359–2381. doi:ten.1007/s00198-014-2794-ii
PubMed Abstract | CrossRef Full Text | Google Scholar
Cummings, South. R., San Martin, J., McClung, M. R., Siris, East. S., Eastell, R., Reid, I. R., et al. (2009). Denosumab for prevention of fractures in postmenopausal women with osteoporosis. Northward. Engl. J. Med. 361 (viii), 756–765. doi:10.1056/NEJMoa0809493
PubMed Abstract | CrossRef Full Text | Google Scholar
Deeks, J. J., Higgins, J. P. T., and Altman, D. K. (2008). "Analyzing information and undertaking meta-analyses," in Cochrane handbook for systematic reviews of interventions 501. Editors J. P Higgins, and Southward. Green (Oxford, UK: The Cochrane Collaboration).
Google Scholar
Diem, Southward. J., Blackwell, T. Fifty., Stone, One thousand. L., Yaffe, M., Haney, E. M., Bliziotes, M. Yard., et al. (2007). Employ of antidepressants and rates of hip bone loss in older women: the study of osteoporotic fractures. Arch. Intern. Med. 167 (12), 1240–1245. doi:x.1001/archinte.167.12.1240
PubMed Abstract | CrossRef Full Text | Google Scholar
Dubois, E. F., Roder, E., Dekhuijzen, P. N., Zwinderman, A. E., and Schweitzer, D. H. (2002). Dual free energy X-ray absorptiometry outcomes in male COPD patients afterward treatment with unlike glucocorticoid regimens. Breast 121 (5), 1456–1463. doi:10.1378/breast.121.5.1456
PubMed Abstruse | CrossRef Full Text | Google Scholar
Ellis, M. Yard., Bone, H. G., Chlebowski, R., Paul, D., Spadafora, S., Smith, J., et al. (2008). Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for nonmetastatic breast cancer. J. Clin. Oncol. 26 (xxx), 4875–4882. doi:10.1200/JCO.2008.16.3832
PubMed Abstract | CrossRef Total Text | Google Scholar
Etminan, Grand., Sadatsafavi, Yard., Ganjizadeh Zavareh, South., Takkouche, B., and FitzGerald, J. M. (2008). Inhaled corticosteroids and the risk of fractures in older adults: a systematic review and meta-assay. Drug Saf. 31 (five), 409–414. doi:x.2165/00002018-200831050-00005
PubMed Abstract | CrossRef Full Text | Google Scholar
Fuller, K., Wong, B., Play tricks, S., Choi, Y., and Chambers, T. J. (1998). TRANCE is necessary and sufficient for osteoblast-mediated activation of bone resorption in osteoclasts. J. Exp. Med. 188 (5), 997–1001. doi:10.1084/jem.188.v.997
PubMed Abstract | CrossRef Total Text | Google Scholar
Gage, B. F., Birman-Deych, E., Radford, M. J., Nilasena, D. South., and Folder, E. F. (2006). Risk of osteoporotic fracture in elderly patients taking warfarin: results from the National Registry of Atrial Fibrillation two. Arch. Intern. Med. 166 (2), 241–246. doi:x.1001/archinte.166.2.241
PubMed Abstract | CrossRef Total Text | Google Scholar
Gnant, M., Pfeiler, G., Dubsky, P. C., Hubalek, M., Greil, R., Jakesz, R., et al. (2015). Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 386 (9992), 433–443. doi:10.1016/S0140-6736(fifteen)60995-3
PubMed Abstract | CrossRef Full Text | Google Scholar
Gu, H. F., Gu, L. J., Wu, Y., Zhao, X. H., Zhang, Q., Xu, Z. R., et al. (2015). Efficacy and safety of denosumab in postmenopausal women with osteoporosis: a meta-analysis. Medicine 94 (44), e1674. doi:x.1097/MD.0000000000001674
PubMed Abstract | CrossRef Full Text | Google Scholar
Hernlund, E., Svedbom, A., Ivergard, M., Compston, J., Cooper, C., Stenmark, J., et al. (2013). Osteoporosis in the European spousal relationship: medical management, epidemiology and economical burden. A report prepared in collaboration with the international osteoporosis foundation (IOF) and the European federation of pharmaceutical industry associations (EFPIA). Arch. Osteoporos 8, 136. doi:ten.1007/s11657-013-0136-1
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hita-Contreras, F., Martínez-López, E., González-Matarín, P., Mendoza, N., Cruz-Díaz, D., Ruiz-Ariza, A., et al. (2014). Association of bone mineral density with postural stability and the fear of falling in Spanish postmenopausal women. Maturitas 79 (3), 322–328. doi:x.1016/j.maturitas.2014.07.015
PubMed Abstract | CrossRef Full Text | Google Scholar
Hofbauer, L. C., and Schoppet, M. (2004). Clinical implications of the osteoprotegerin/RANKL/RANK organization for bone and vascular diseases. Jama 292 (iv), 490–495. doi:10.1001/jama.292.4.490
PubMed Abstract | CrossRef Full Text | Google Scholar
Holick, M. F., Binkley, N. C., Bischoff-Ferrari, H. A., Gordon, C. M., Hanley, D. A., Heaney, R. P., et al. (2011). Evaluation, handling, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96 (7), 1911–1930. doi:10.1210/jc.2011-0385
PubMed Abstruse | CrossRef Total Text | Google Scholar
Institute for Clinical Systems Improvement (ICSI) (2017). Diagnosis and treatment of osteoporosis. 9th Edn. Bloomington: Institute for Clinical Systems Comeback (ICSI).
Jadad, A. R., Moore, R. A., Carroll, D., Jenkinson, C., Reynolds, D. J., Gavaghan, D. J., et al. (1996). Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control. Clin. Trials 17 (1), i–12. doi:x.1016/0197-2456(95)00134-4
PubMed Abstract | CrossRef Full Text | Google Scholar
Khalili, H., Huang, E. S., Jacobson, B. C., Camargo, C. A., Feskanich, D., and Chan, A. T. (2012). Employ of proton pump inhibitors and risk of hip fracture in relation to dietary and lifestyle factors: a prospective accomplice study. BMJ 344, e372. doi:10.1136/bmj.e372
PubMed Abstract | CrossRef Total Text | Google Scholar
Koh, J. M., Chung, D. J., Chung, Y. S., Kang, One thousand. I., Kim, I. J., Min, Y. M., et al. (2016). Assessment of denosumab in Korean postmenopausal women with osteoporosis: randomized, double-blind, placebo-controlled trial with open-label extension. Yonsei Med. J. 57 (4), 905–914. doi:10.3349/ymj.2016.57.four.905
PubMed Abstruse | CrossRef Full Text | Google Scholar
Kumagai, Y., Hasunuma, T., and Padhi, D. (2011). A randomized, double-blind, placebo-controlled, single-dose study to evaluate the prophylactic, tolerability, pharmacokinetics and pharmacodynamics of denosumab administered subcutaneously to postmenopausal Japanese women. Bone 49 (five), 1101–1107. doi:x.1016/j.bone.2011.08.007
PubMed Abstruse | CrossRef Full Text | Google Scholar
Lacey, D. L., Tan, H. L., Lu, J., Kaufman, S., Van, Chiliad., Qiu, West., et al. (2000). Osteoprotegerin ligand modulates murine osteoclast survival in vitro and in vivo. Am. J. Pathol. 157 (2), 435–448. doi:ten.1016/S0002-9440(ten)64556-seven
PubMed Abstract | CrossRef Full Text | Google Scholar
Lacey, D. Fifty., Timms, E., Tan, H. L., Kelley, K. J., Dunstan, C. R., Burgess, T., et al. (1998). Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93 (2), 165–176. doi:ten.1016/s0092-8674(00)81569-ten
PubMed Abstract | CrossRef Full Text | Google Scholar
Lee, J., Lee, S., Jang, S., and Ryu, O. H. (2013). Age-related changes in the prevalence of osteoporosis according to gender and skeletal site: the korea national health and nutrition examination survey 2008-2010. Endocrinol. Metab. (Seoul) 28 (3), 180–191. doi:10.3803/EnM.2013.28.3.180
PubMed Abstruse | CrossRef Full Text | Google Scholar
Lewiecki, E. Thou., Miller, P. D., McClung, Grand. R., Cohen, S. B., Bolognese, M. A., Liu, Y., et al. (2007). Two-twelvemonth treatment with denosumab (AMG 162) in a randomized phase 2 study of postmenopausal women with low BMD. J. Bone Miner Res. 22 (12), 1832–1841. doi:ten.1359/jbmr.070809
PubMed Abstract | CrossRef Total Text | Google Scholar
Loke, Y. Yard., Cavallazzi, R., and Singh, Due south. (2011). Risk of fractures with inhaled corticosteroids in COPD: systematic review and meta-analysis of randomised controlled trials and observational studies. Thorax 66 (8), 699–708. doi:10.1136/thx.2011.160028
PubMed Abstruse | CrossRef Full Text | Google Scholar
Majumdar, S. R., Josse, R. G., Lin, M., and Eurich, D. T. (2016). Does sitagliptin affect the rate of osteoporotic fractures in type 2 diabetes? Population-based cohort report. J. Clin. Endocrinol. Metab. 101 (5), 1963–1969. doi:x.1210/jc.2015-4180
PubMed Abstract | CrossRef Total Text | Google Scholar
National Library of Medicine (2010). Management of osteoporosis in postmenopausal women: 2010 position statement of the N American Menopause Society. Menopause 17 (1), 25–54. doi:10.1097/gme.0b013e3181c617e6
PubMed Abstract | CrossRef Full Text | Google Scholar
McClung, 1000. R., Lewiecki, Due east. G., Cohen, S. B., Bolognese, G. A., Woodson, G. C., Moffett, A. H., et al. (2006). Denosumab in postmenopausal women with low os mineral density. New Engl. J. Med. 354 (eight), 821–831. doi:10.1056/NEJMoa044459
PubMed Abstract | CrossRef Total Text | Google Scholar
Moher, D., Liberati, A., Tetzlaff, J., and Altman, D. G. (2009). Preferred reporting items for systematic reviews and meta-analyses: the PRISMA argument. PLoS Med. six (7), e1000097. doi:10.1371/journal.pmed.1000097
PubMed Abstract | CrossRef Full Text | Google Scholar
Murad, G. H., Chu, H., Lin, 50., and Wang, Z. (2018). The consequence of publication bias magnitude and direction on the certainty in prove. BMJ Evid. Based Med. 23 (iii), 84–86. doi:x.1136/bmjebm-2018-110891
PubMed Abstract | CrossRef Full Text | Google Scholar
Nakamura, T., Matsumoto, T., Sugimoto, T., Hosoi, T., Miki, T., Gorai, I., et al. (2014). Clinical Trials Express: fracture adventure reduction with denosumab in Japanese postmenopausal women and men with osteoporosis: denosumab fracture intervention randomized placebo controlled trial (DIRECT). J. Clin. Endocrinol. Metab. 99 (vii), 2599–2607. doi:10.1210/jc.2013-4175
PubMed Abstract | CrossRef Total Text | Google Scholar
Nakamura, T., Matsumoto, T., Sugimoto, T., and Shiraki, Thousand. (2012). Dose-response study of denosumab on bone mineral density and bone turnover markers in Japanese postmenopausal women with osteoporosis. Osteoporos. Int. 23 (3), 1131–1140. doi:x.1007/s00198-011-1786-8
PubMed Abstruse | CrossRef Full Text | Google Scholar
Ngamruengphong, Southward., Leontiadis, 1000. I., Radhi, S., Dentino, A., and Nugent, K. (2011). Proton pump inhibitors and risk of fracture: a systematic review and meta-analysis of observational studies. Am. J. Gastroenterol. 106 (seven), 1209–1219. doi:10.1038/ajg.2011.113
PubMed Abstract | CrossRef Full Text | Google Scholar
Pedroza-Tobías, A. (1999). Assessing the influence of a single written report in meta-assay. Stata Tech. Bull. 47, 15–17.
Google Scholar
Qaseem, A., Forciea, Yard. A., McLean, R. M., and Denberg, T. D. (2017). Clinical guidelines commission of the American College of P. Treatment of low bone density or osteoporosis to prevent fractures in men and women: a clinical practise guideline update from the American College of physicians. Ann. Intern. Med. 166 (eleven), 818–839. doi:x.7326/M15-1361
PubMed Abstract | CrossRef Full Text | Google Scholar
Richards, J. B., Papaioannou, A., Adachi, J. D., Joseph, L., Whitson, H. E., Prior, J. C., et al. (2007). Effect of selective serotonin reuptake inhibitors on the take a chance of fracture. Arch. Intern. Med. 167 (2), 188–194. doi:ten.1001/archinte.167.two.188
PubMed Abstract | CrossRef Full Text | Google Scholar
Sambrook, P., and Cooper, C. (2006). Osteoporosis. Osteoporos. Lancet (London, England) 367 (9527), 2010–2018. doi:10.1016/s0140-6736(06)68891-0
CrossRef Total Text | Google Scholar
Seeman, E., Delmas, P. D., Hanley, D. A., Sellmeyer, D., Cheung, A. G., Shane, East., et al. (2010). Microarchitectural deterioration of cortical and trabecular bone: differing effects of denosumab and alendronate. J. Bone Miner Res. 25 (8), 1886–1894. doi:10.1002/jbmr.81
PubMed Abstract | CrossRef Full Text | Google Scholar
Sharma, Southward., Fraser, Yard., Lovell, F., Reece, A., and McLellan, A. R. (2008). Characteristics of males over fifty years who nowadays with a fracture: epidemiology and underlying adventure factors. J. Bone Jt. Surg Br 90 (1), 72–77. doi:10.1302/0301-620X.90B1.18773
CrossRef Full Text | Google Scholar
Simonet, W. Southward., Lacey, D. L., Dunstan, C. R., Kelley, Chiliad., Chang, Thou. S., Luthy, R., et al. (1997). Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89 (2), 309–319. doi:x.1016/s0092-8674(00)80209-iii
PubMed Abstract | CrossRef Full Text | Google Scholar
Tit, D. G., Bungau, Southward., Iovan, C., Nistor Cseppento, D. C., Endres, L., Sava, C., et al. (2018). Effects of the hormone replacement therapy and of soy isoflavones on bone resorption in postmenopause. J. Clin. Med. 7 (10), 297. doi:10.3390/jcm7100297
CrossRef Full Text | Google Scholar
Tit, D. Thousand., Pallag, A., Iovan, C., Furau, Thou., Furau, C., and Bungau, S. (2017). Somatic-vegetative symptoms evolution in postmenopausal women treated with phytoestrogens and hormone replacement therapy. Iran J. Public Health 46 (11), 1528–1534.
PubMed AbstractGoogle Scholar
von Keyserlingk, C., Hopkins, R., Anastasilakis, A., Toulis, M., Goeree, R., Tarride, J. E., et al. (2011). Clinical efficacy and safety of denosumab in postmenopausal women with low bone mineral density and osteoporosis: a meta-analysis. Semin. Arthritis Rheum. 41 (two), 178–186. doi:10.1016/j.semarthrit.2011.03.005
PubMed Abstruse | CrossRef Full Text | Google Scholar
Yasuda, H., Shima, N., Nakagawa, N., Yamaguchi, K., Kinosaki, One thousand., Mochizuki, S., et al. (1998). Osteoclast differentiation gene is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. UsaA. 95 (vii), 3597–3602. doi:10.1073/pnas.95.7.3597
PubMed Abstract | CrossRef Full Text | Google Scholar
Zhou, Z., Chen, C., Zhang, J., Ji, Ten., Liu, Fifty., Zhang, G., et al. (2014). Safety of denosumab in postmenopausal women with osteoporosis or low bone mineral density: a meta-analysis. Int. J. Clin. Exp. Pathol. 7 (5), 2113–2122.
PubMed AbstractGoogle Scholar
Source: https://www.frontiersin.org/articles/10.3389/fphar.2021.588095/full
0 Response to "Efficacy and Safety of Denosumab for the Treatment of Osteoporosis a Systematic Review"
Post a Comment